What our Studies Showed
Lasix ONYU received Tentative Approval from the FDA in October 2024. Lasix ONYU is not approved for sale.
Two studies were reported in 2023 by the University of Glasgow.
Goals of the Studies
- Compare plasma levels and diurectic response of subcutaneous infusion of the new formulation with standard furosemide adminstered intravenously (IV) including the following measures:
- Bioavailability (amount of drug that enters the body)
- Pharmacodynamic parameters (action of the drug on the body)
- Diuresis (amount of urine output)
- Natriuresis (amount of sodium excreted in urine)
- Pharmacokinetics (plasma furosemide levels over time)
- Identify any adverse events, including infusion site pain, skin irritation, or device failure associated with the novel treatment.
- Understand patient acceptability of the new treatment
Summary of Results
- The novel subcutaneous furosemide formulation infused over 5 hours resulted in complete bioavailability (amount of drug that enters the body) when compared to the same dose of furosemide administered IV as a single dose.
. - Subcutaneous furosemide resulted in similar diuresis (amount of urine output) and natriuresis (amount of sodium excreted) when compared to IV furosemide
. - Using the Lasix ONYU Infusor was feasible and well-tolerated by patients
Details of Results
BIOAVAILABILITY
Bioavailability (amount of drug that enters the body) of subcutaneous infusion versus IV furosemide was 112%.
Furosemide given by single IV bolus dose is rapidly excreted with the first voids and hence the slightly lower number for IV.
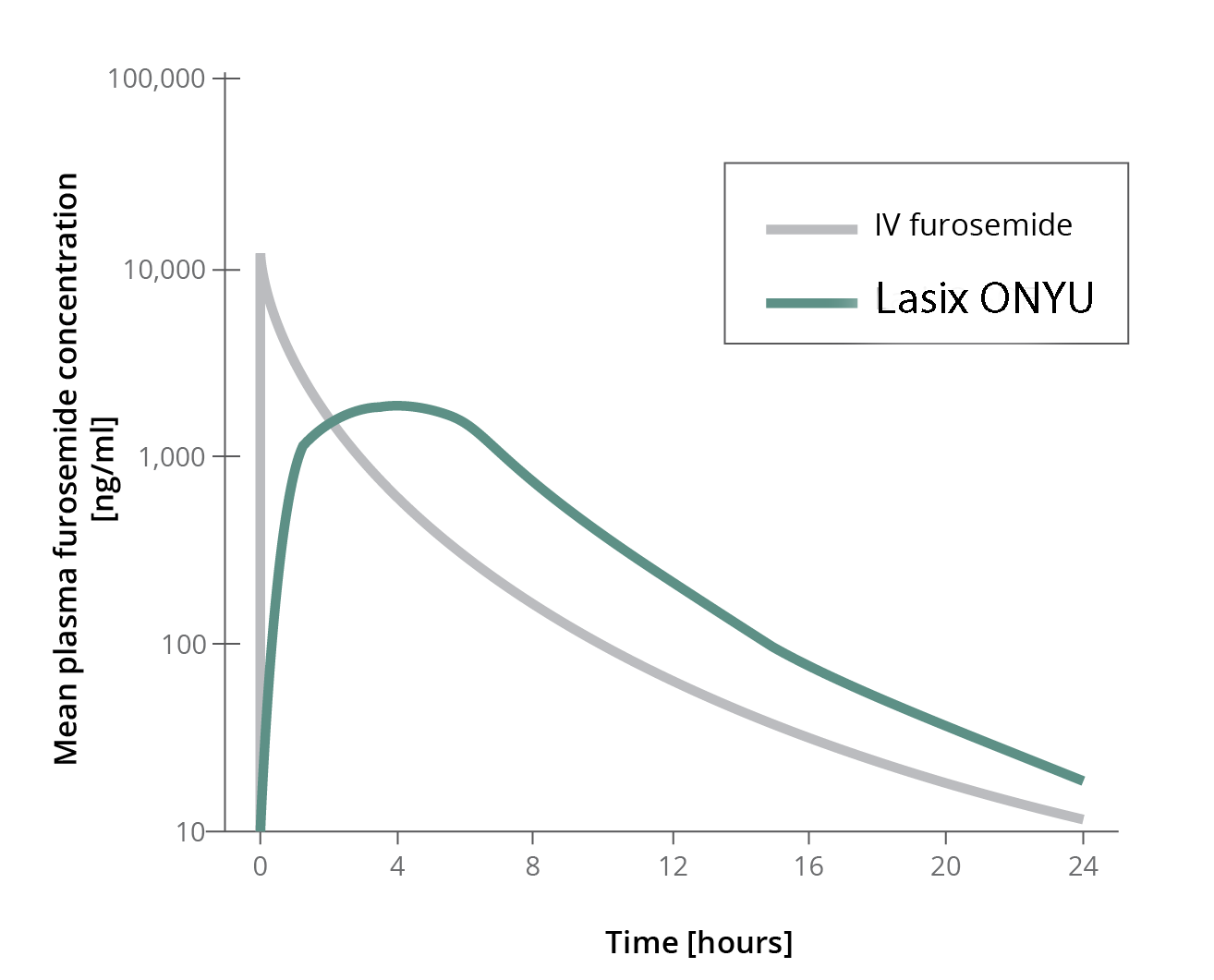
PHARMACODYNAMIC PARAMETERS
Pharmacodynamic parameters describe the action of the drug on the body. The study looked at two related pharmacodynamic parameters.
Subcutaneous furosemide resulted in similar results in terms of diuresis (amount of urine output) and natriuresis (amount of sodium in urine) when compared to IV furosemide. On average subcutaneous furosemide resulted in 3.5L diuresis over 24hrs when compared to 3.0L with IV.
Diuresis is more spread-out with the slow infusion when compared to IV bolus. It starts slightly later but lasts considerably longer. This tempered diuretic was selected to give a patient a more comfortable experience while preserving the full diuretic response.
Diuresis (amount of urine output)
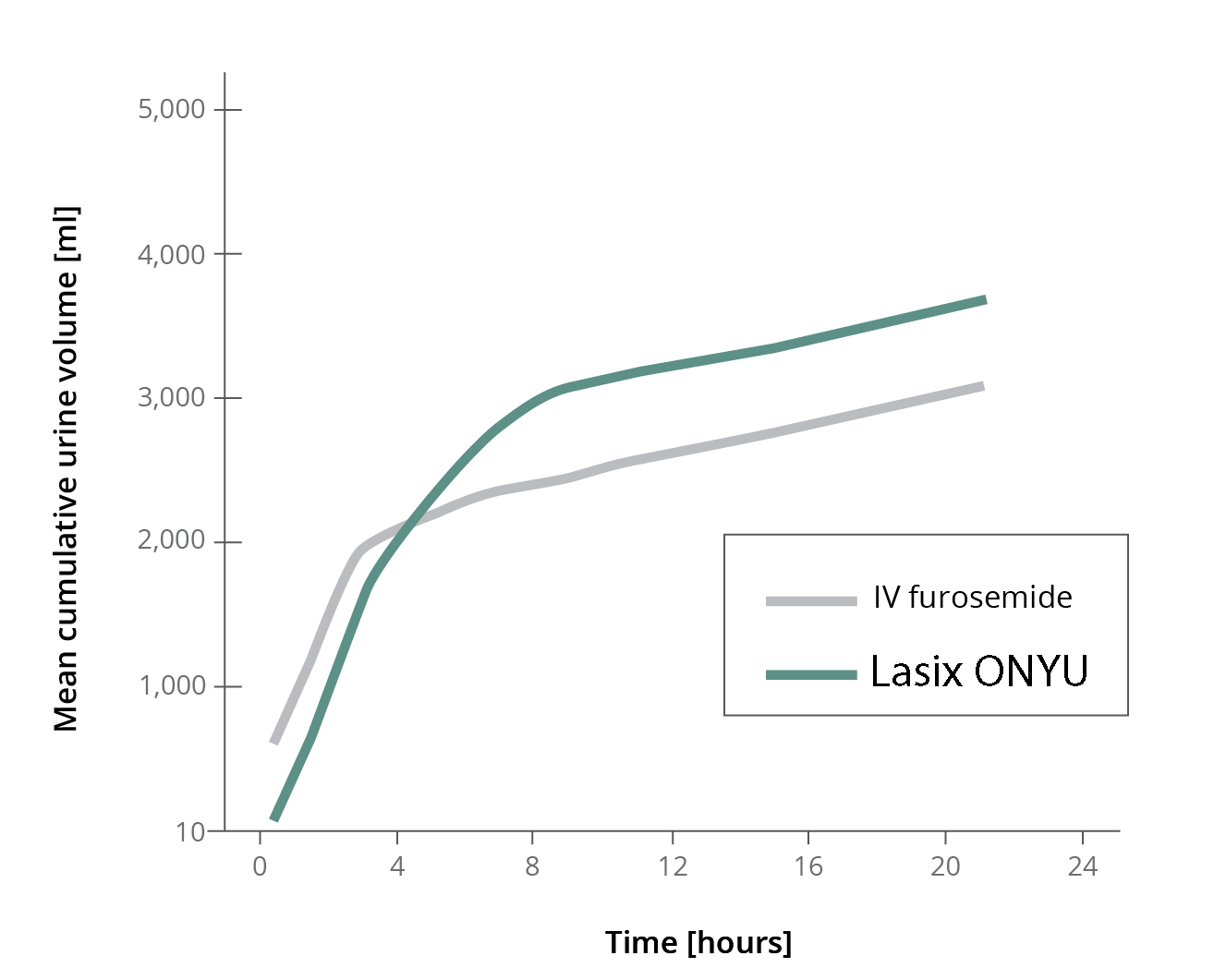
Natriuresis (amount of sodium excreted)

PHARMACOKINETICS
Pharmacokinetics is the activity of drugs in the body over time, including the processes by which drugs are absorbed, distributed in the body, localized in the tissues, and excreted.
The plasma furosemide time curves show the expected differences:
-
- IV bolus results in a very high peak value that is about 7 times higher than the average maximum level with the 5hr infusion
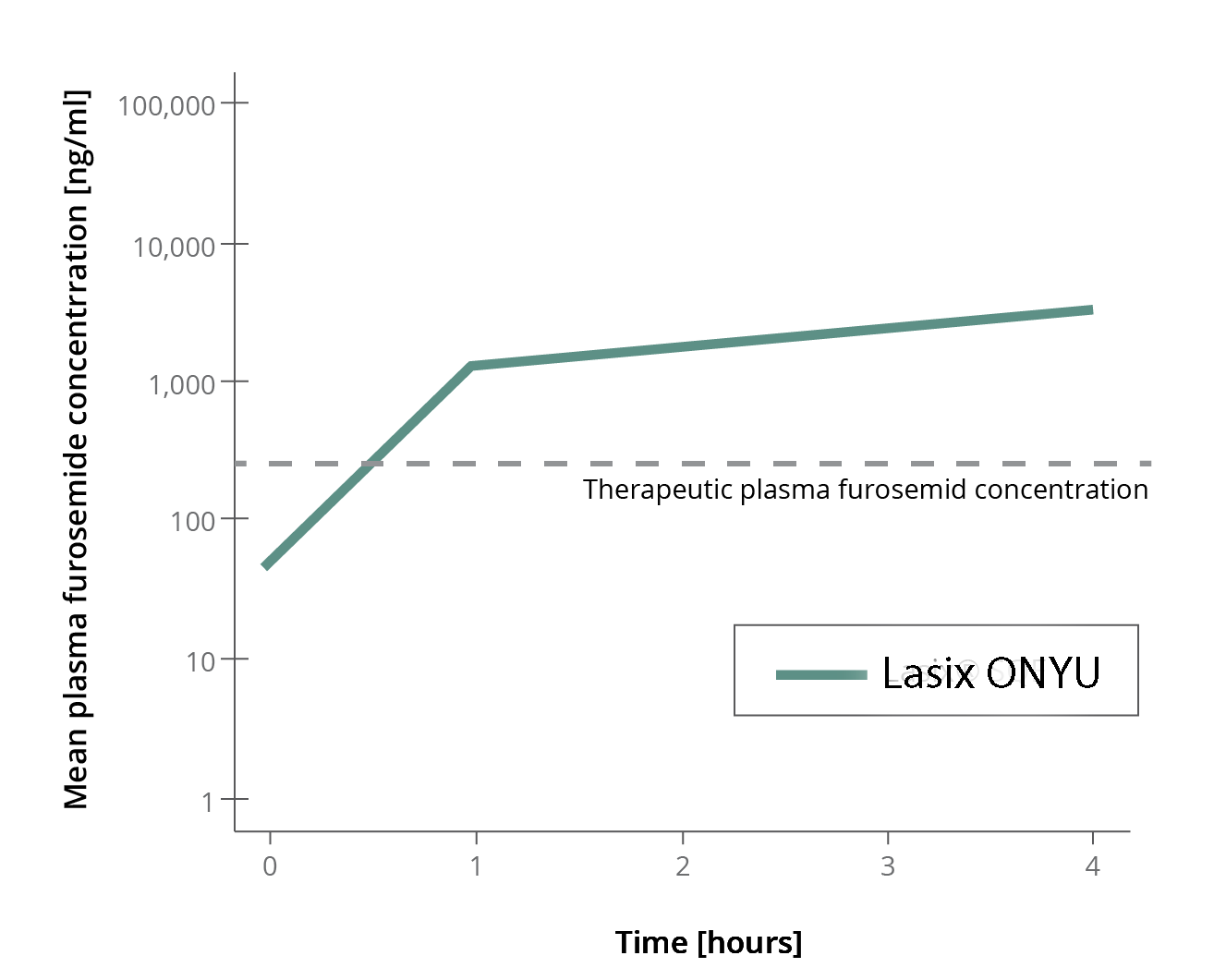
- Plasma levels with the Infusor reach levels sufficient for diuresis in less than 1hr
- Effective plasma levels are maintained for more than 6 hours
The second study conducted at the University of Glasgow showed that the Infusor in hospitalized heart failure patients performed well. Plasma levels at 1 and 4 hours after start resulted in similar plasma levels as the first study.
This study showed that use of the special Infusor results in furseomide plasma levels sufficient to cause a marked diurectic response in patients with fluid overload.
ADVERSE EFECTS
- Events: No treatment related adverse events were identified.
- Infusion Site Pain: 95% of the participants reported no or minimal pain.
- Skin irritation: 4 out of 20 participants had minor, transient skin reaction.
- Adhesion: The device adhered well in 19 of the 20 participants. One Infusor came loose after approximately 1 hour and 25 mins. This was attributed to profuse sweating which prevents the adhesive from bonding to the skin.
PATIENT ACCEPTABILITY
The Infusor was determined to have excellent usability.
The median System Usability Scale score was 99 (where 100 was a maximal score); 14 (70%) participants scored above 85.
This product is not approved for sale. Efficacy and safety of the new product have been established and labeling and manufacturing requirements have also been met.
Up Next
A large randomised controlled trial, co-sponsored by NHS (National Health Service) and the University of Glasgow was started in Q4 of 2022 in the UK . This trial investigates the effectiveness and safety of use use of the new treatment at home compared to usual care strategy with IV furosemide delivered in hospital. Patients hospitalised with worsening heart failure are randomized shortly after their admission to the novel home-base treatment or usual hospital care.
Sign up for our mailing list to stay informed about clinical studies with our investigational products and the results.
